
Institut für Organische Chemie, Universität Regensburg, 93040 Regensburg, Germany
 |
Susanne Kümmel, R. Lechner, and Burkhard König
Institut für Organische Chemie, Universität Regensburg, 93040 Regensburg, Germany |
 |
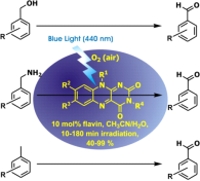
Flavins are nature's beloved redox co-factors.[1] When irradiated by blue light, their ability to oxidize is dramatically increased and thus they can be used in photocatalysis (see figure).[2] They are easily accessible by synthesis and their properties can be tuned by substitution pattern (i.e. by changing R1-4 in the figure).[3]
Recently we reported different organic reactions with vitamin B2 derivatives as efficient photocatalysts.[4,5] The key step of the photocatalytic cycle is the formation of the triplet state of the flavin.[6] Based on the conclusions of these investigations we are now able to present some improved derivatives of this catalyst that yield higher triplet state populations due to heavy atom effect. These new photocatalysts accelerate the oxidation reactions and improve quantum yields.
Research supports: Deutsche Bundesumweltstiftung (DBU), DFG: GRK 1626 - Chemical Photocatalysis