
Department of Structural Biology, Max-Planck-Institute of Biophysics, Max-von-Laue-Straße 3, 60438 Frankfurt, Germany
 |
Werner Kühlbrandt
Department of Structural Biology, Max-Planck-Institute of Biophysics, Max-von-Laue-Straße 3, 60438 Frankfurt, Germany |
 |
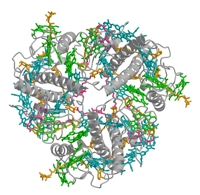
We use X-ray or electron crystallography, single-particle electron cryo-microscopy and electron cryo-tomography to study the structure, function and supramolecular organisation of membrane proteins. The plant light-harvesting complex LHC-II is one of the most abundant membrane proteins on Earth. It collects most of the solar energy in the biosphere, and is the prototype of a highly conserved family of membrane proteins that fuels plant photosynthesis. In addition, LHC-II plays an important role in the organization of the thylakoid membrane, the structure of the photosynthetic apparatus, the regulation of energy flow between the two photosystems, and in the controlled dissipation of excess excitation energy under light stress. Based on the atomic structure of LHC-II and an alignment of the remarkably well-engineered and highly conserved LHC polypeptides, we identify several key features that are likely to be present in all members of the LHC family.
The in situ arrangement of membrane protein complexes in chloroplast thylakoids and plant mitochondria was examined by electron cryo-tomography. Together with the high-resolution structure of LHC-II, this provides us with a molecular model of membrane interaction in chloroplast grana. In addition, we found that the chloroplast ATP synthase is monomeric, and confined to membrane regions of minimal curvature. By contrast, the mitochondrial ATP synthase forms long rows of dimers at the position of highest curvature in the inner membrane cristae. The strikingly different arrangement of the ATP synthase in chloroplasts and mitochondria correlates with differences in membrane potential and pH gradient.