
Buchmann Institute for Molecular Life Sciences and Institute of Biochemistry, Goethe University, Max von Laue Straße 15, D-60438 Frankfurt/Main, Germany
e-mail: a.gottschalk@em.uni-frankfurt.de
 |
Alexander Gottschalk
Buchmann Institute for Molecular Life Sciences and Institute of Biochemistry, Goethe University, Max von Laue Straße 15, D-60438 Frankfurt/Main, Germany e-mail: a.gottschalk@em.uni-frankfurt.de |
 |
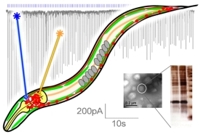
Nervous system function relies on the transmission of chemical signals between neurons, and on functional integration of many neurons within circuits that determine the generation of a behavior, e.g. in response to external signals. We study these elementary processes in the nematode C. elegans, whose nervous system contains just 302, likely "hard-wired" neurons. The animal is transparent, allowing applications of optical methods, and all synapses between its neurons have been physically mapped by electron microscopy. This lays the foundations for a possible complete understanding of also the functional connectivity of this nervous system, and how it generates behaviors. However, first sufficient information about the role of individual neurons and the nature of their synaptic connections (inhibitory, excitatory, modulatory, synaptic strength) has to be acquired. We use microbial rhodopsins (e.g. channel- or, halorhodopsin, as well as the proton pumps Arch or Mac) to photo-activate or inhibit individual neurons in live animals, probing their influence on behavior quantitatively. We established methods to express rhodopsins in single neurons, or to illuminate individual neurons in freely moving animals, and to analyze behavioral alterations we evoke. Several nodes of the same neuronal network can be probed simultaneously, and rhodopsin-based photostimulation can further be combined with imaging of neuronal activity. Using genetics, we probe how functionality of individual neurons depends on the proteins they express.
To analyze basic processes of synaptic transmission, we use photostimulation of motor neurons, combined with genetic manipulation and electrophysiology, and using also behavioral effects as readout. With Stefan Eimer (ENI, Göttingen), we analyze morphological effects of hyperstimulation of neurons, probing processes involved in synapse homeostasis and recovery. Neurons in whole animals are photostimulated, then they are rapidly cryo-fixated and synaptic ultrastructure is analyzed by electron microscopy, in a time dependent manner. Morphological changes at the synapse are correlated with phenotypes as analyzed by electrophysiology. I will present select examples of the methods we developed, and the applications we use them for.